INVESTIGATIVE REPORT: Health Canada and the Pfizer-BioNTech COMIRNATY Deception
How the Government of Canada misled the Canadians on Regulatory Status of the Pfizer-BioNTech Comirnaty Covid-19 Vaccine as "Safe and Effective"
This is Part One of a three part Investigative Report, as follows:
Part One:
Reviews published claims by Health Canada that the Pfizer-BioNTech COMIRNATY Covid-19 vaccine has been “approved” and “proven safe and effective”, and shows that the Government knows these claims are untrue, by
Providing details of the Health Canada - Regulatory Decision Summary - to authorize the use of the Pfizer-BioNTech COMIRNATY Covid-19 vaccine;
Outlining specifics on the regulatory classification assigned by the Minister of Justice to the Pfizer-BioNTech COMIRNATY Covid-19 vaccine; then
Provides the definition of “Fraud” in the Criminal Code of Canada, and
Presents an analysis conducted by researchers at Columbia University on “COVID vaccination and age-stratified all-cause mortality risk”.
Part Two and Part Three of this Investigative Report have been published separately.
Part Two: documents how Canadian regulators may rely on the US FDA (“the FDA”) as a “foreign regulatory authority” in the Pre-positioning of Designated COVID-19 Drugs, and outlines the content and implications of two (2) confusing letters the FDA wrote to Pfizer Inc., and to BioNTech GmbH on August 23, 2021, with respect to the authorization (re: BioNTech & COMIRNATY) and licensing (re: COMIRNATY) of their Covid-19 vaccines.
Part Three: Epidemiology and the Politics of Deceit, provides a overview of the known Epidemiology of Covid-19.
Bait and Switch? Division 8 of the Food and Drug Regulations — New Drugs
As with its essentially identical (while legally distinct - see Part Two) predecessor BioNTech, the Pfizer-BioNTech COMIRNATY Covid-19 vaccine is only "authorized" (def: to grant permission) for use in Canada.
In fact, this Covid-19 vaccine has not been approved (def: to sanction officially), nor has it been established that it is safe and effective.
A manufacturer of an (emergency) authorized drug is protected by a blanket liability shield, but loses that liability shield once the drug is approved (see Part Two).
This has significant implications for parents of children under age 19, as well for employers who have imposed vaccine mandates on workers; who may have relied on Health Canada’s published claims that it had '‘approved” the COMIRNATY Covid-19 vaccine.
For details, see the following three (3) screenshots and related documents found in the hyper-links.
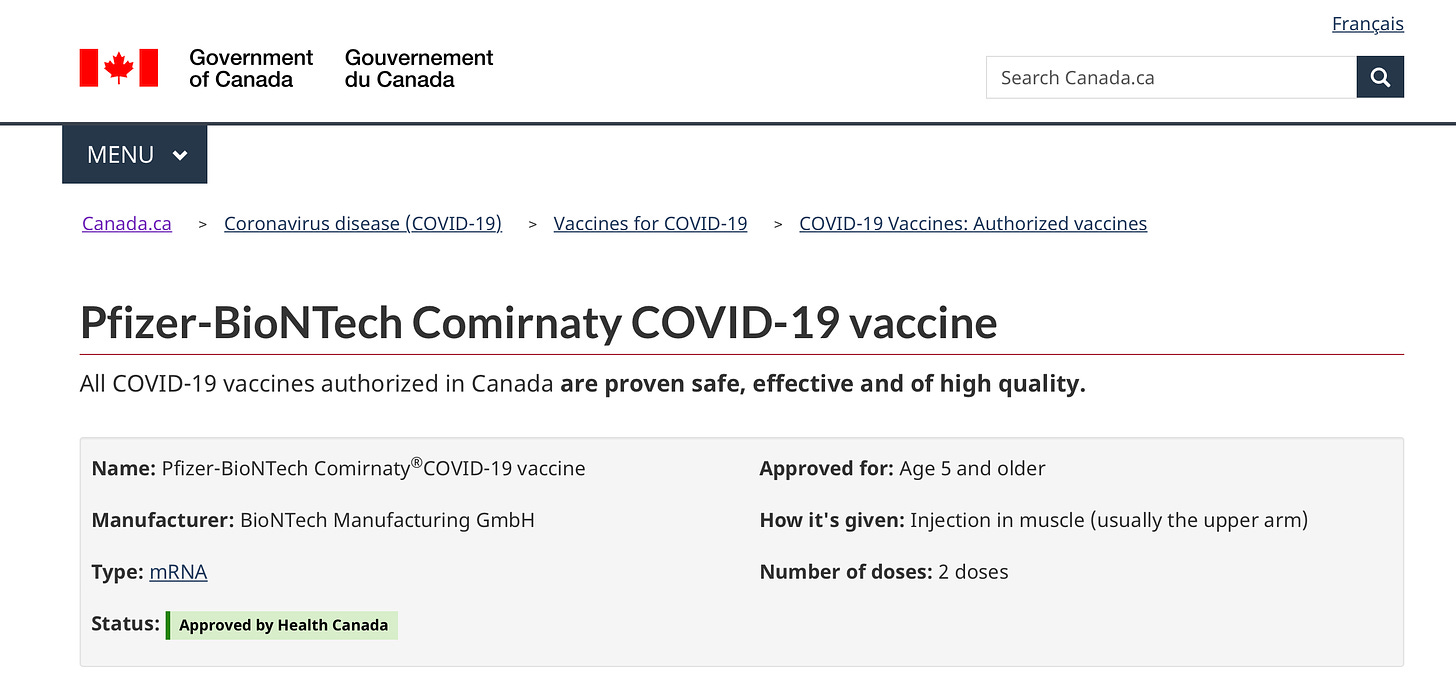
1) Below is a screenshot of a Health Canada webpage concerning the Pfizer-BioNTech COMIRNATY COVID-19 vaccine, wherein Health Canada declares that:
All COVID-19 vaccines authorized in Canada "“are proven safe and effective”,
The Status of the Pfizer-BioNTech COMIRNATY COVID-19 is: “Approved by Health Canada”, and
It has been “Approved for: Age 5 and older”.
Figure 1: Government of Canada “ALL COVID-19 vaccines in Canada are proven safe, effective”
2) However, in the screenshot shown below taken from Health Canada’s Regulatory Decision Summary - COMIRNATY — we can clearly see that it states that this Covid-19 vaccine has (only) been “authorized”, under and in accordance with an obscure section of the ten-part 1,154 page, Food and Drug Regulations – Division 8.
To what does Division 8 of the Food and Drug Regulations relate? Read on.
Figure 2: Regulatory Decision Summary - COMIRNATY - Health Canada (Division 8)
3) Shown below are screenshots of Part C, Division 8 of the Food and Drug Regulations – New Drugs, where it states that this classification means a drug:
(a) … that has not been sold as a drug in Canada for sufficient time and in sufficient quantity to establish in Canada the safety and effectiveness of that substance for use as a drug.
Therefore, by its very definition, a ‘New Drug’ is a drug that has not yet been shown to be safe and effective.
Figure 3: Division 8 of the Food and Drug Regulations - New Drugs
Division 8 of the Food and Drug Regulations goes on to define a “designated Covid-19 Drug" as:
a new drug for which the purpose and conditions of use recommended by the manufacturer relate to COVID-19;
drogue désignée contre la COVID-19 Drogue nouvelle dont les fins et le mode d’emploi recommandés par le fabricant ont trait à la COVID-19.
Health Canada’s published claims that the Pfizer-BioNTech COMIRNATY COVID-19 vaccine has been “approved” and “proven safe and effective” are therefor in direct conflict with the drugs actual regulatory status.
In fact, we can clearly see that the fizer-BioNTech COMIRNATY COVID-19 vaccine to have been only authorized for use secondary to its classification under Division 8 of the Food and Drug Regulation as a “New Drug” — meaning, by definition, that the safety and efficacy of COMIRNATY has not been established. To claim otherwise elsewhere would appear to be misleading, at best.
Can governments in Canada mandate or otherwise coerce the uptake of a drug when its safety and efficacy has not been established? No, they most certainly cannot, and the liability implications could be staggering.
So what is really going on? Have Canadians been misled so that Proof of Vaccination edicts could be plausibly deployed by the Provinces, vaccine mandates imposed by a great many large employers, and this New Drug even marketed directly to children? If so, was this done in order to increase the uptake of an unproven drug by deploying deceptive and manipulative techniques recommended by the World Health Organization on an unwary public?
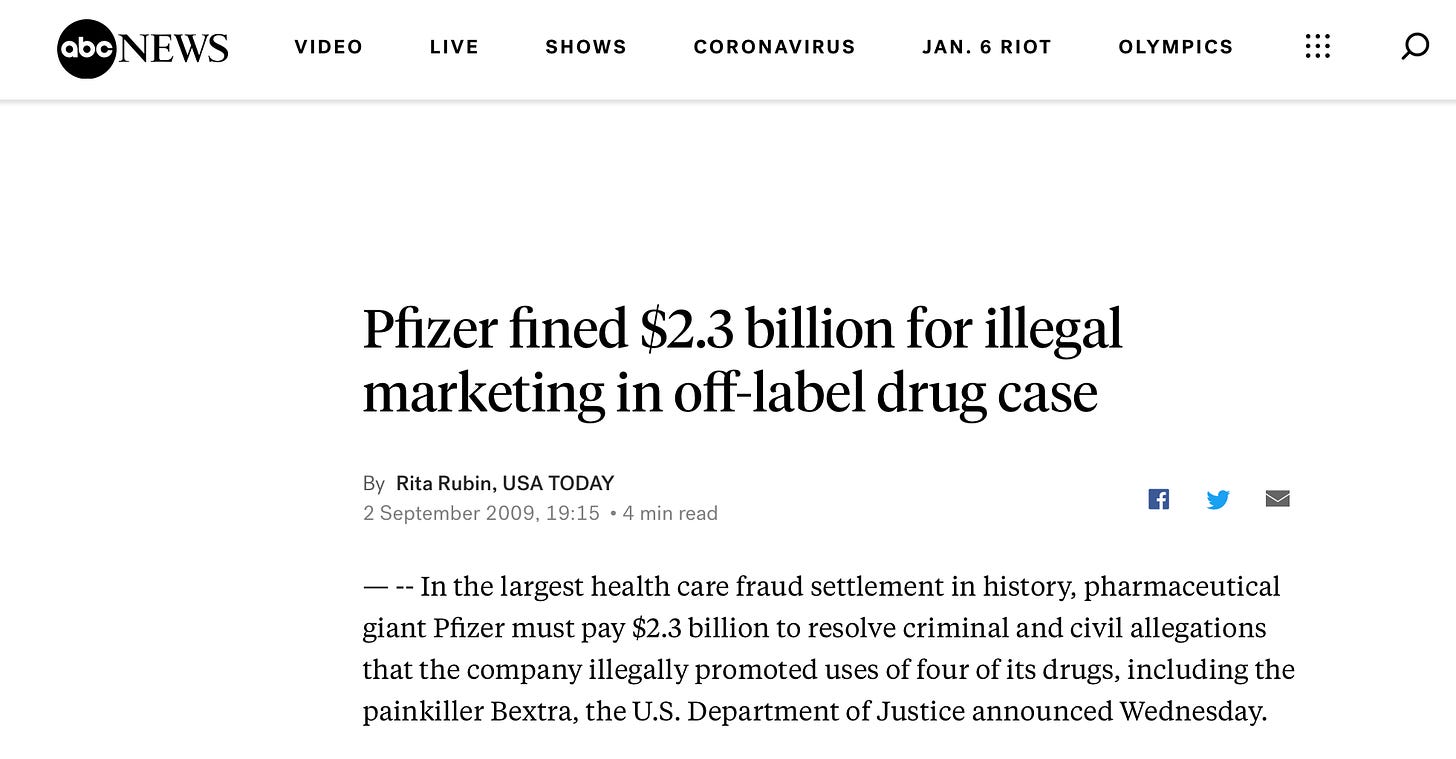
If we were to attempt to unravel these questions we might begin by asking: would Pfizer’s liability shield (see Part Two) be at risk if fraud is found? After all, fraud, for which Big Pharma has plead guilty in the past, eviscerates all contracts.
Pfizer itself has paid out billions in civil lawsuits, including $2.3 billion that Pfizer and a subsidiary agreed to pay out for the largest healthcare fraud settlement in the history of the US Justice Department.
Section 380 of the Criminal Code of Canada — Fraud, states:
(1) Every one who, by deceit, falsehood or other fraudulent means, whether or not it is a false pretence within the meaning of this Act, defrauds the public or any person, whether ascertained or not, of any property, money or valuable security or any service,
(a) is guilty of an indictable offence and liable to a term of imprisonment not exceeding fourteen years, where the subject-matter of the offence is a testamentary instrument or the value of the subject-matter of the offence exceeds five thousand dollars; or
(2) Affecting public market
Every one who, by deceit, falsehood or other fraudulent means, whether or not it is a false pretence within the meaning of this Act, with intent to defraud, affects the public market price of stocks, shares, merchandise or anything that is offered for sale to the public is guilty of an indictable offence and liable to imprisonment for a term not exceeding fourteen years.
Pfizer’s own trial data for its Covid-19 vaccine, which is starting to be released in response to a Freedom of Information Act (FOIA) request to the FDA, do nothing to assuage safety concerns. Quite the contrary. We have learned that, cumulatively, between December 1, 2020, and February 28, 2021 — a period of just 2.5 months — Pfizer received 42,086 injury reports, including 1,223 fatalities.1
This is an especially salient issue given that the FDA is currently in a US Federal Court (which Pfizer attempted to join) trying to prevent a group of public health officials and medical professionals from having further access to the FDA's Pfizer Covid-19 vaccine licensing records via FOIA. An issue that the senior editor of the British Medical Journal (BMJ) weighed-in on in a recent editorial: Covid-19 vaccines and treatments: we must have raw data, now (for a detailed review, see end note 2).
Here’s another riddle to consider, a report from Reuters on February 5, 2022 — Pfizer drops India vaccine application after regulator seeks local trial — stated that the drug company would rather miss out on billions of dollars of guaranteed profit selling its mRNA Covid-19 vaccines in India than submit to an independent local trial to:
determine if the vaccine is SAFE and generates an immune response in its citizens.
Let this sink in.
Perhaps Pfizer and the FDA are hiding safety data of much more significance than is generally known? If so, a number of recent studies may provide some clues about what is being hidden.
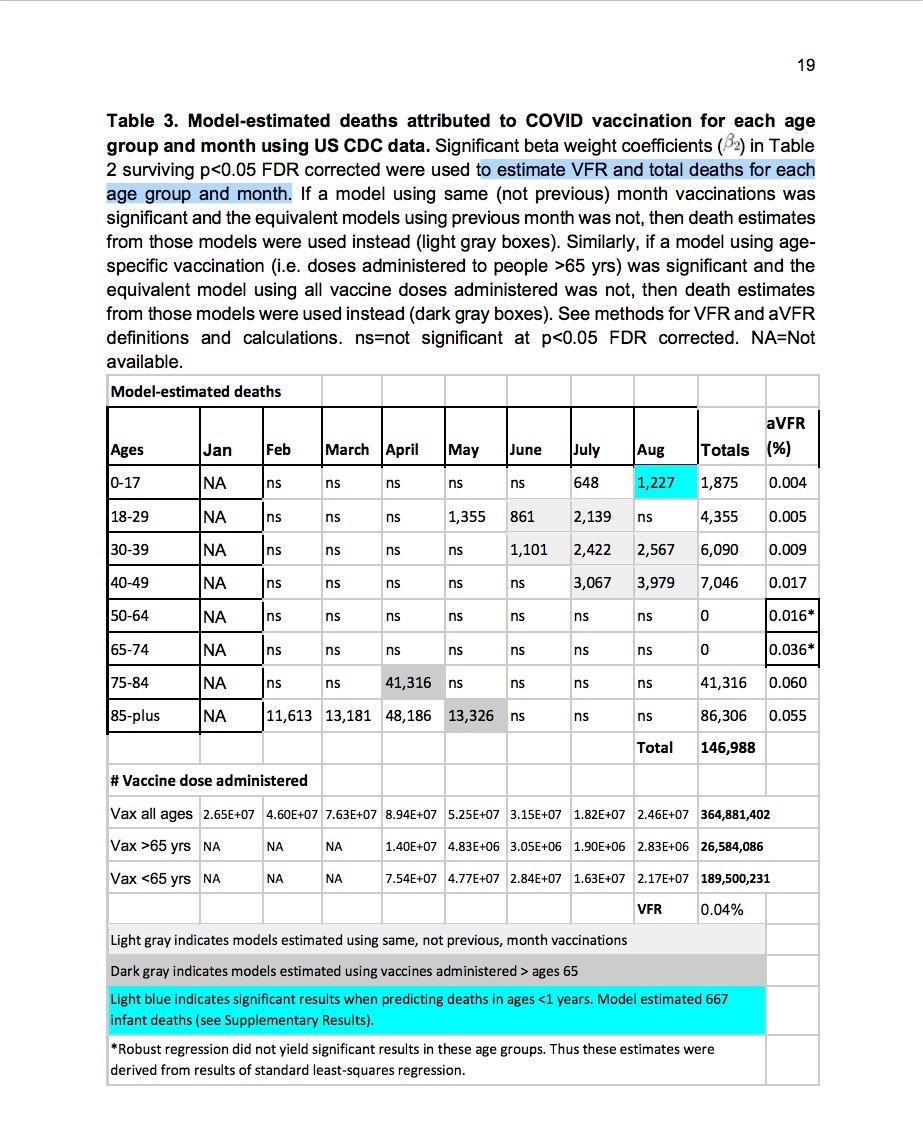
In an analysis published in October 2021 by researchers at Columbia University — COVID vaccination and age-stratified all-cause mortality risk — the authors stated:
Accurate estimates of COVID vaccine-induced severe adverse event and death rates are critical for risk-benefit ratio analyses of vaccination and boosters against SARS-CoV-2 coronavirus in different age groups. However, existing surveillance studies are not designed to reliably estimate life-threatening events or vaccine-induced fatality rates (VFR).
According to the authors:
Comparing our age-stratified VFRs [vaccine-induced fatality rates] with published age-stratified coronavirus infection fatality rates (IFR) suggests the risks of COVID vaccines and boosters outweigh the benefits in children, young adults and older adults with low occupational risk or previous coronavirus exposure.
We discuss implications for public health policies related to boosters, school and workplace mandates, and the urgent need to identify, develop and disseminate diagnostics and treatments for life-altering vaccine injuries.
If “existing surveillance studies are not designed to reliably estimate life-threatening events or vaccine-induced fatality rates”, is it not disingenuous for public health officials to claim that “no safety issues were detected” with respect to the administration of these New Drugs?
See below “Model-estimated deaths attributed to Covid Vaccination” in the US for Jan-Aug 2021, from the analysis conducted by researchers at Columbia University. Please, note the highlighted boxes and the Totals column.
Figure 4: from COVID vaccination and age-stratified all-cause mortality risk (October 2021)
Meanwhile, Sweden has decided against recommending COVID vaccines for children aged 5-11, arguing that the benefits did not outweigh the risks. Not so Health Canada.
"With the knowledge we have today, with a low risk for serious disease for kids, we don't see any clear benefit with vaccinating them," a Sweden Health Agency official told a news conference.
As noted in the preamble, in Part Two of this Investigative Report — The FDA, a foreign regulatory authority in Canada? — we will review the section of Division 8 of the Food and Drug Regulations that allows Health Canada, in the Pre-positioning of Designated COVID-19 Drugs, to rely on:
an application (that) has been submitted to a foreign regulatory authority to authorize the sale of the designated COVID-19 drug.
Which will be followed by a detailed analysis of the FDA’s authorization and licensing of the COMIRNATY Covid-19 vaccine, in response to a submission by Pfizer-BioNTech of a Biologic License Application (BLA) for this Covid-19 drug.
End Notes:
1 My dictionary defines “established” in the present context as: shown to be true or certain by determining the facts, and “proven” as: demonstrated by evidence to be true. In other words, these are synonyms; they mean the same thing.
2 Re: BMJ https://doi.org/10.1136/bmj.o102: Covid-19 vaccines and treatments: we must have raw data, now
The British Medical Journal (BMJ) is a weekly peer-reviewed medical trade journal published by the trade union of the British Medical Association, and has editorial freedom from the British Medical Association. In an editorial published on January 19, 2022 the BMJ called for the release of the raw data from the trials used to first authorize the Covid-19 vaccines for (emergency) use, and now mandate vaccine uptake.
Excerpt from the BMJ editorial:
“Today, despite the global rollout of COVID-19 vaccines and treatments, the anonymized participant-level data underlying the trials (used by governments to first authorize the use of the vaccines and now increasingly mandate their uptake) for these new products remain inaccessible to doctors, researchers, and the public—and are likely to remain that way for years to come,”
“This is morally indefensible for all trials, but especially for those involving major public health interventions.”
BMJ also accused pharmaceutical companies of “reaping vast profits without adequate independent scrutiny of their scientific claims,” pointing to Pfizer, whose COVID vaccine trial was “funded by the company and designed, run, analyzed, and authored by Pfizer employees.”
New York-headquartered Pfizer still holds that trial data and has indicated that it won’t begin considering requests for such data until May 2025 – 24 months after the primary study completion date of May 15, 2023, which is listed on ClinicalTrials.gov as well as in Part Two of this report.
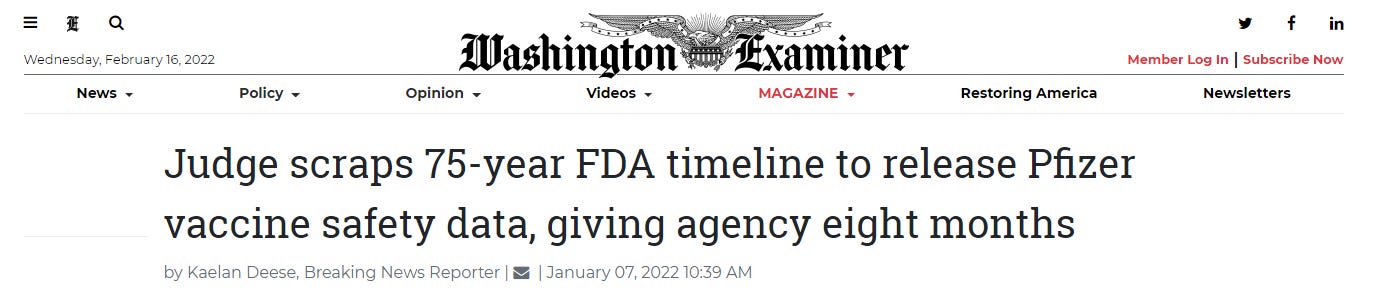
Meanwhile, The Food and Drug Administration (FDA) had asked a judge to give it 75 years to produce all the data concerning the Pfizer and BioNTech vaccine.
However, a judge recently ordered that the FDA make public 12,000 pages of the data it used to make decisions regarding approvals for the Pfizer/BioNTech COVID-19 vaccine by the end of the month. The FDA must also release Pfizer’s vaccine data at a rate of 55,000 pages a month until all of the requested pages are public.
“We are left with publications but no access to the underlying data on reasonable request,” BMJ said.
“This is worrying for trial participants, researchers, clinicians, journal editors, policymakers, and the public. The journals that have published these primary studies may argue that they faced an awkward dilemma, caught between making the summary findings available quickly and upholding the best ethical values that support timely access to underlying data. In our view, there is no dilemma; the anonymized individual participant data from clinical trials must be made available for independent scrutiny.”
BMJ added that regulators are not there to “dance to the tune of rich global corporations and enrich them further” but to protect the general public’s health and for that reason, they said, we need “complete data transparency for all studies, we need it in the public interest, and we need it now.”
Also of note (H/T Kelly Brown @rubiconcapital), Pfizer recently added new and peculiar items deep in its business risk disclosures re: clinical trial data, in its Q4 earnings report for 2021, concerning: “...risks associated with...further information regarding the quality of pre-clinical, clinical or safety data, including by audit or inspection;”
And "...challenges related to public confidence or awareness of our COVID-19 vaccine … including … CONCERNS ABOUT CLINICAL DATA INTEGRITY and prescriber and pharmacy education;"
“Fact Checkers” noted that Pfizer didn’t determine cause and effect, only that more than 1,000 vaccinated people died in the first three months after the vaccine's emergency use authorization from the FDA on December 11, 2020.
Pfizer’s document spells out its limitations:
Pfizer's safety database contains cases of AEs (Adverse Events) reported spontaneously to Pfizer, cases reported by the health authorities, cases published in the medical literature, cases from Pfizer-sponsored marketing programs, non-interventional studies, and cases of serious AEs reported from clinical studies regardless of causality assessment.
What we don’t know about the “cause and effect” of the Adverse Event’s reported post-vaccination is precisely why researchers require Pfizer’s raw data. “Just trust us” won’t do.